Feb 1, 2026 Product
MedGuard-AI: Visual Inspection for Medical Packaging Integrity and Compliance
An enterprise-grade vision system that ensures zero-defect pharmaceutical and medical device packaging by detecting seal, label, and structural defects within fully auditable, regulatory-compliant workflows.
Prompt Input: "Inspect the current lot of sterilized syringes for seal integrity and label accuracy using the high-resolution feed from Camera A. Cross-reference the printed expiration date and batch number with the master production record. Identify any micro-fractures or seal folds in the secondary packaging and generate an immutable audit trail compliant with FDA standards."
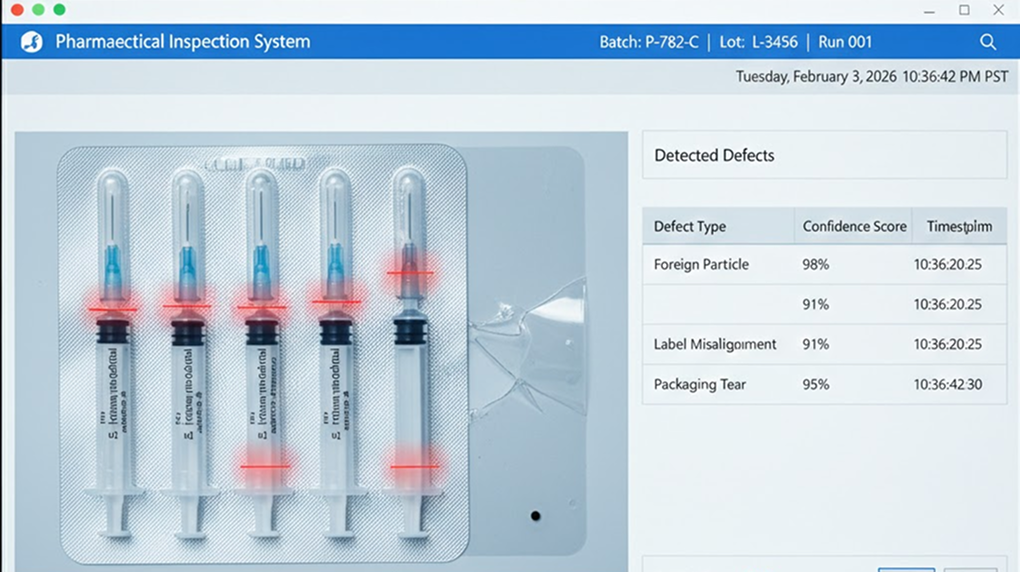
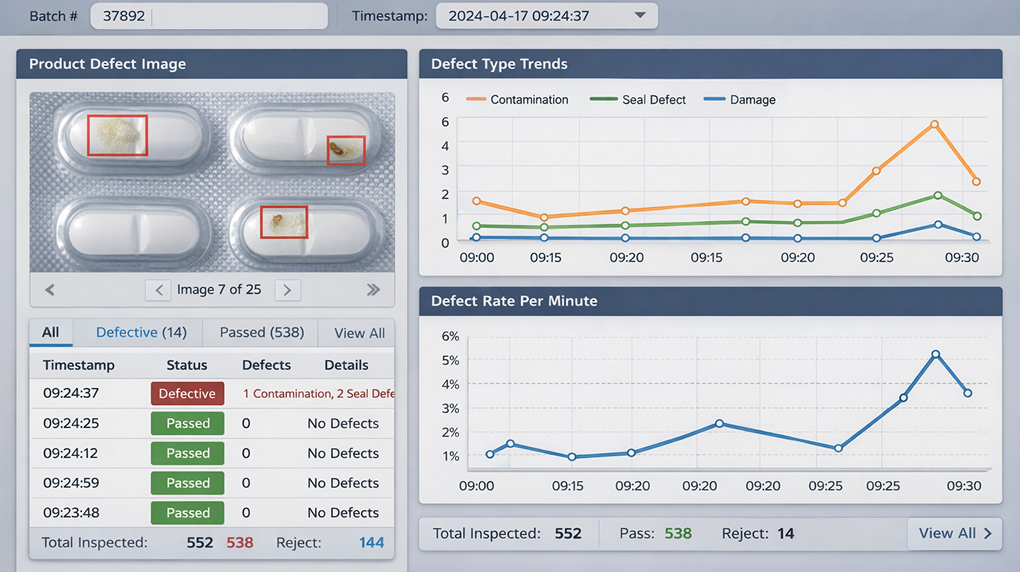

Note: The live demo regenerates dynamically. Prompts and interface elements may differ from the screenshots to showcase the system's full capabilities.
Result:
A production-ready software system featuring:
1. Deep learning models for high-accuracy visual defect classification (e.g., cracks, folds, and contamination);
2. An automated compliance-reporting engine for regulatory submissions;
3. Real-time visualization of inspection results and defect trends;
4. A seamless data logging layer that records every pass/fail decision with timestamps for audit purposes.